Agentic AI Transforming ESG for Pharmaceutical Generics
Pharmaceutical generics manufacturers face a critical juncture as ESG performance becomes mandatory due to regulatory pressures, commercial demands, and operational opportunities. Leading companies are leveraging AI-powered ESG platforms to gain competitive advantages, while those who delay risk falling behind. Agentic AI, a system of specialized AI models, offers a transformative solution for optimizing ESG operations and achieving real-time, prescriptive outcomes. This article discusses “The Ai-ESG Imperative for Pharmaceutical Generics: How Agentics Ai-ESG is transforming ESG from a compliance burden into structural competitive advantage — and why the window to act is closing fast?”
Posted by
Ai-ESG Desk
Posted at
AI Transformation
Posted on
The Ai-ESG Imperative for Pharmaceutical Generics
There is a moment in every industry's evolution when a new operating paradigm shifts from optional to obligatory. For pharmaceutical generics manufacturers, that moment has arrived — and it concerns ESG (environmental, social, and governance) performance.
This is not a story about compliance checklists or sustainability reports gathering dust in boardroom filing cabinets. This is a story about competitive architecture, market access, and the irreversible convergence of three powerful forces: regulation, capital, and customer expectation.
The analogy is ERP in the 1990s: companies that automated financial management first gained decade-long structural advantages. Ai-ESG is that same inflection point — happening right now, in the generics sector.
The manufacturers who build an AI-native ESG capability today will not merely comply, they will use sustainability as a lever to win hospital tenders, reduce operating costs, access green financing, and build supply chains that are genuinely resilient. Those who wait will spend the next decade playing expensive catch-up.
Section 01 - THE BURNING PLATFORM
Why Is ESG Now Mandatory for Pharmaceutical Generics Manufacturers?
ESG is now mandatory for pharmaceutical generics manufacturers due to three converging pressures:
(1) EU CSRD legislation mandating full Scope 1-3 disclosure from FY2024,
(2) Hospital and public sector procurement frameworks adding ESG scoring to supplier evaluation criteria
(3) Global parent group ESG commitments cascading reporting obligations to EU subsidiaries.
For most of the past decade, ESG in pharma was a voluntary, reputation-driven exercise. That era is over. Three concurrent pressures have permanently transformed the landscape:
Regulatory Pressure: CSRD and Framework Compliance
The EU's Corporate Sustainability Reporting Directive (CSRD) is now mandatory from FY2024 for large EU companies, with mid-sized subsidiaries entering scope from FY2025. CDP, EcoVadis, and GRI disclosures are increasingly required for public hospital tender qualification across EU markets. Penalties for non-compliance are material and escalating.
Commercial Pressure: Hospital Procurement ESG Scoring
Hospital procurement frameworks across Portugal and the EU are actively adding ESG scoring criteria to supplier evaluations. EcoVadis Silver is becoming a minimum threshold for many hospital Group Purchasing Organisations (GPOs). This is not a future risk — contracts are being awarded and lost on ESG performance today.
Operational Opportunity: Hidden Value in ESG Data
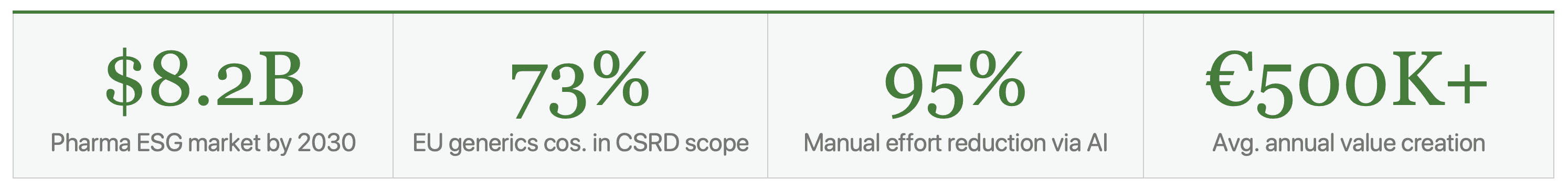
The data behind the compliance obligations represents a previously untapped goldmine of operational insight. Manufacturing facilities burning 15–20% more energy than necessary. Supply chains carrying €100K–€300K+ of avoidable Scope 3 costs annually. Logistics networks that could route 25% more efficiently. ESG data, properly captured and intelligently applied, is a direct profit lever.

Section 02 - INDUSTRY BENCHMARKS
What Are Leading Pharmaceutical Generics Companies Doing for ESG?
Leading generics companies including Teva, Sandoz, Stada, Dr. Reddy's, and Aurobindo are deploying AI-powered ESG platforms that deliver real-time Scope 1-3 tracking, automated multi-framework reporting, supplier carbon scoring, and circular packaging intelligence.
Results include 23–50% improvement across key metrics within 12–24 months of deployment.
Analysis of publicly available ESG reports, CDP disclosures, and EcoVadis ratings across 2022–2025 reveals a clear pattern: the companies gaining the most from ESG investment are those treating it as an operational intelligence programme — not a reporting obligation.
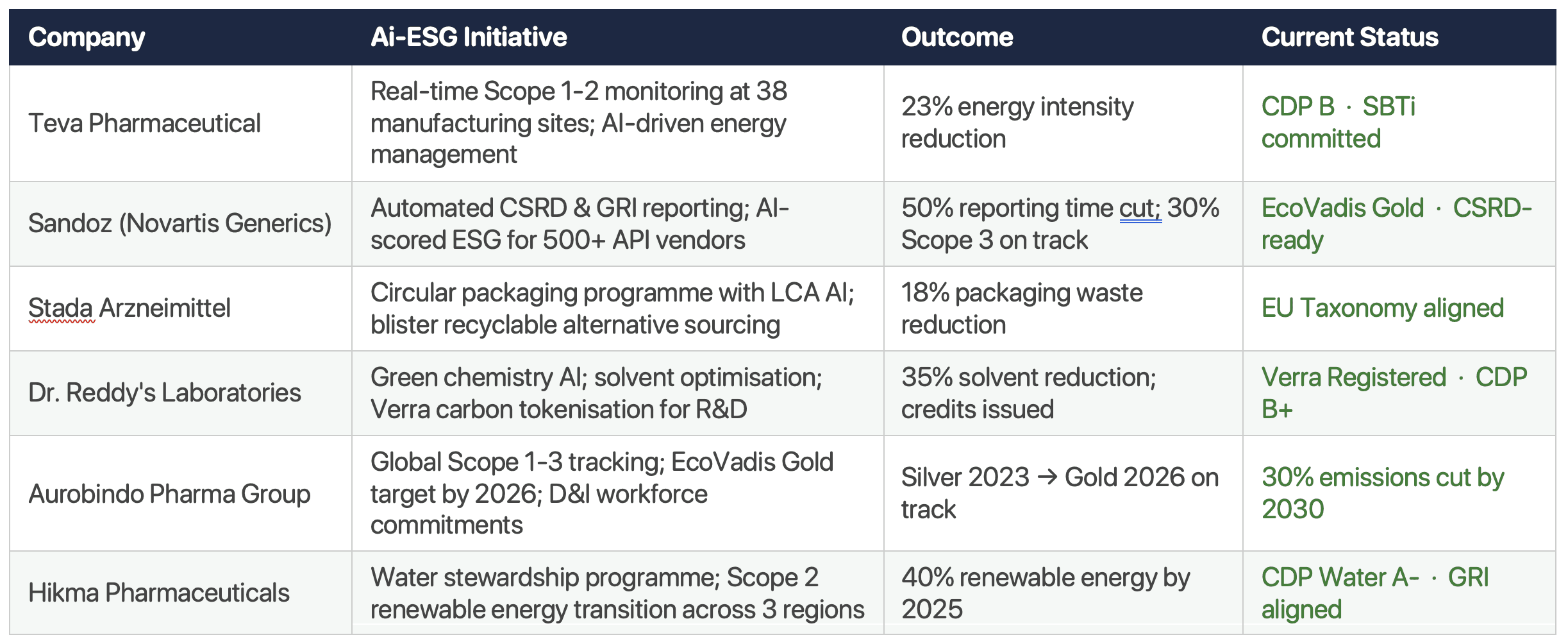
Sources: Publicly available ESG reports, CDP disclosures, and EcoVadis ratings. Data: FY2022–2025. Agentics research.
Section 03 - THE MATURITY GAP
What Is the ESG Maturity Gap in Pharmaceutical Generics and Why Does It Matter?
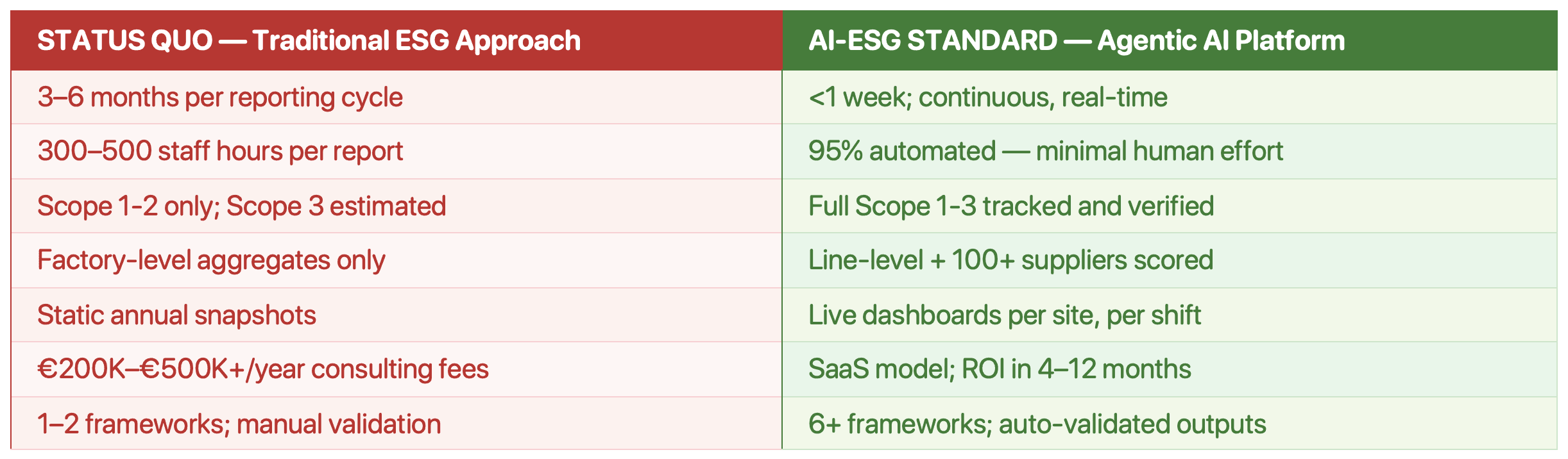
The ESG maturity gap refers to the structural difference between how most mid-size generics manufacturers currently manage ESG (manually, periodically, with limited Scope 3 visibility) versus the AI-ESG standard now set by market leaders (real-time, automated, covering full Scope 1-3).
This gap creates direct exposure in hospital procurement, investor relations, and regulatory compliance.
Most generics manufacturers today rely on one of two approaches: periodic consultant-led reporting cycles, or first-generation ESG software tools that automate data entry but deliver little else. Neither is adequate for the scale, complexity, or speed of what CSRD and commercial buyers now require.
Section 04 - AGENTIC AI ARCHITECTURE
What Is Agentic AI and Why Is It the Right Architecture for Pharma ESG?
Agentic AI refers to systems of specialised, goal-directed AI models (agents) that can perceive their environment, reason about it, take actions, and learn continuously. For pharma ESG, this means agents that actively optimise operations — reducing energy use, scoring suppliers, generating CSRD reports — rather than passively displaying data or waiting for human instruction.

The Four-Layer Architecture
Layer 1 — DATA: ERPs, IoT sensors, supplier portals, billing systems, and regulatory databases. All connected via standard APIs. No rip-and-replace infrastructure required.
Layer 2 — LLM + RAG: Large Language Models augmented with Retrieval-Augmented Generation for real-time access to regulatory frameworks, emissions factors, IPCC pathway data, and best-practice libraries — updated continuously.
Layer 3 — AGENT LAYER: 100+ specialised vertical agents, each an expert in one domain. The CSRD Reporting Agent knows the directive in granular detail. The Scope 3 Supplier Agent understands API chemistry and packaging LCA. The Green Chemistry Agent is trained on Process Mass Intensity (PMI) metrics. Critically, agents share data — the Scope 3 Agent feeds the CSRD Agent automatically.
Layer 4 — INSIGHT & ACTION: Prescriptive recommendations executed in real time — route changes, supplier flags, energy scheduling. Not dashboards for humans to act on; decisions, with humans in the loop for oversight.

Section 05 - INDUSTRY USE CASES
What Are the Key Ai-ESG Use Cases for Pharmaceutical Generics Manufacturers?
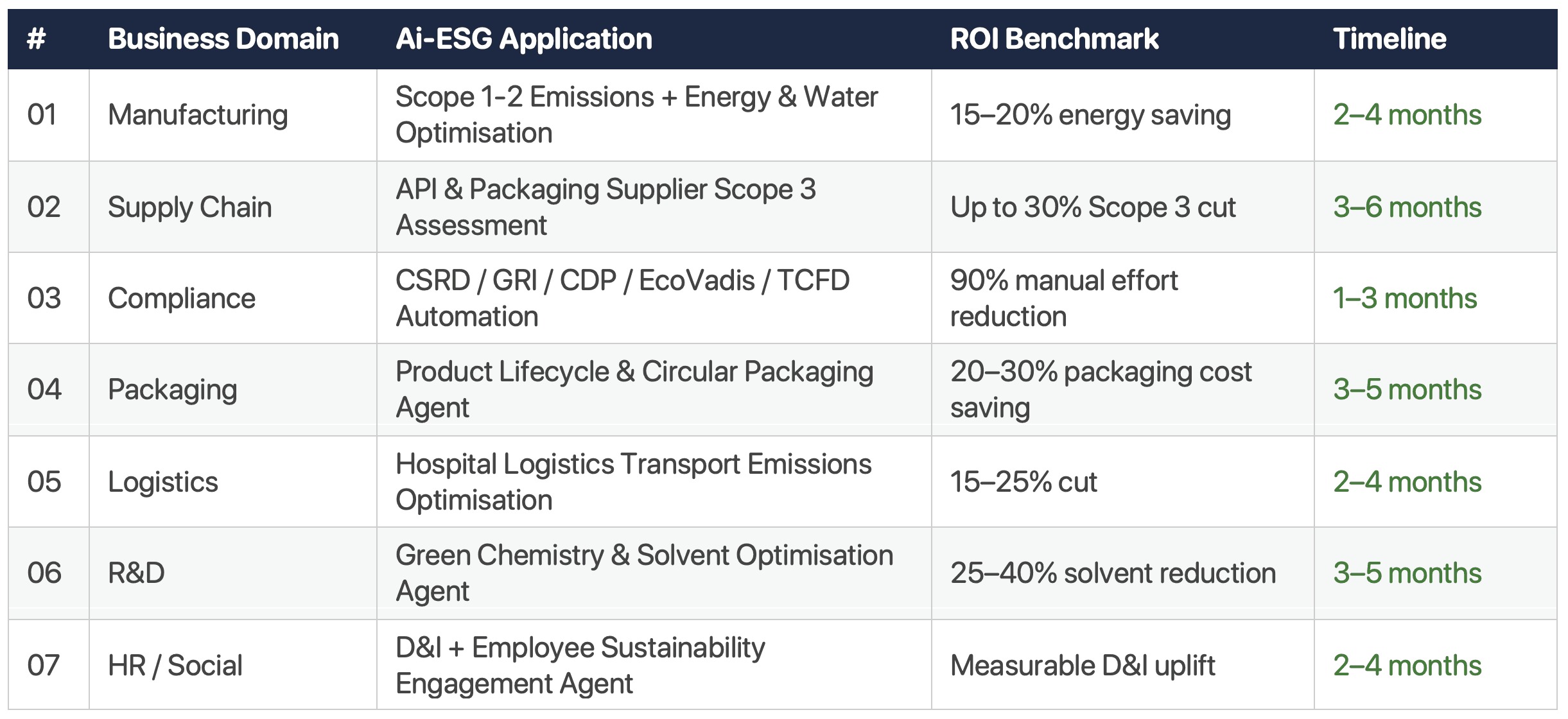
There are 7 high-value Ai-ESG applications mapped to the pharmaceutical generics value chain: (1) manufacturing Scope 1-2 optimisation, (2) API/packaging Scope 3 supply chain, (3) CSRD/regulatory automation, (4) circular packaging LCA, (5) hospital logistics emissions, (6) green chemistry R&D, and (7) D&I workforce analytics. Combined ROI potential: €500K–€2M+ annually.
Deep Dive: Manufacturing Scope 1-2 Emissions Optimisation
High-volume tablet and capsule manufacturing is among the most energy-intensive processes in the pharmaceutical value chain. Boilers, HVAC systems, sterilisation cycles, and clean-in-place (CIP) processes collectively represent 60–75% of a facility's total carbon footprint — and are highly optimisable with real-time AI.
The Manufacturing Emissions Agent operates in three modes simultaneously:
• Monitor mode: Real-time Scope 1-2 measurements per production line and per shift, replacing manual meter reads entirely.
• Optimise mode: Predictive scheduling to align energy-intensive processes with periods of low grid carbon intensity and off-peak pricing, reducing both emissions and energy costs.
• Report mode: Auto-generated kWh-per-unit metrics in exact format required by CSRD, CDP, and group ESG disclosures — zero manual effort.

Deep Dive: Scope 3 API and Packaging Supply Chain
For most pharmaceutical generics manufacturers, purchased goods and services — primarily API and excipient inputs — represent over 72% of total Scope 3 emissions. Yet this is the category with the least visibility and the highest potential for intervention.
A typical mid-size generics manufacturer sources from 100+ API and packaging suppliers spanning India, China, Europe, and the US. The emissions intensity of these supply chains varies enormously — and most manufacturers have no systematic way to identify which suppliers are driving the most carbon exposure, or what procurement or logistics changes would reduce it most efficiently.
The Scope 3 Supplier Agent addresses this by:
• Auto-scoring all API and packaging suppliers on ESG performance using public and proprietary data
• Calculating per-supplier emissions intensity and flagging high-carbon vendors for renegotiation or substitution
• Recommending sea vs. air freight modal shifts with real-time carbon cost comparisons
• Generating audit-ready Scope 3 Category 1 & 4 disclosures for CSRD, CDP, and GRI

Section 06 - ROI & BUSINESS CASE
What Is the ROI of an Ai-ESG Platform for a Pharmaceutical Generics Manufacturer?
A mid-size pharmaceutical generics manufacturer deploying the Agentics Ai-ESG platform can expect €500,000–€2,000,000+ in annual value creation across energy savings, reporting cost reduction, supply chain optimisation, logistics efficiency, and packaging improvements. Payback periods range from 1–12 months depending on the initiative, with manufacturing energy optimisation typically returning investment in 4–8 months.

Note: Figures based on benchmarks from comparable pharmaceutical and generics manufacturing deployments. A company-specific ROI model is produced during the Paid ESG Discovery Workshop using actual operational data.
Section 07 - IMPLEMENTATION ROADMAP
How Long Does It Take to Implement an Ai-ESG Platform for Pharmaceutical Generics?
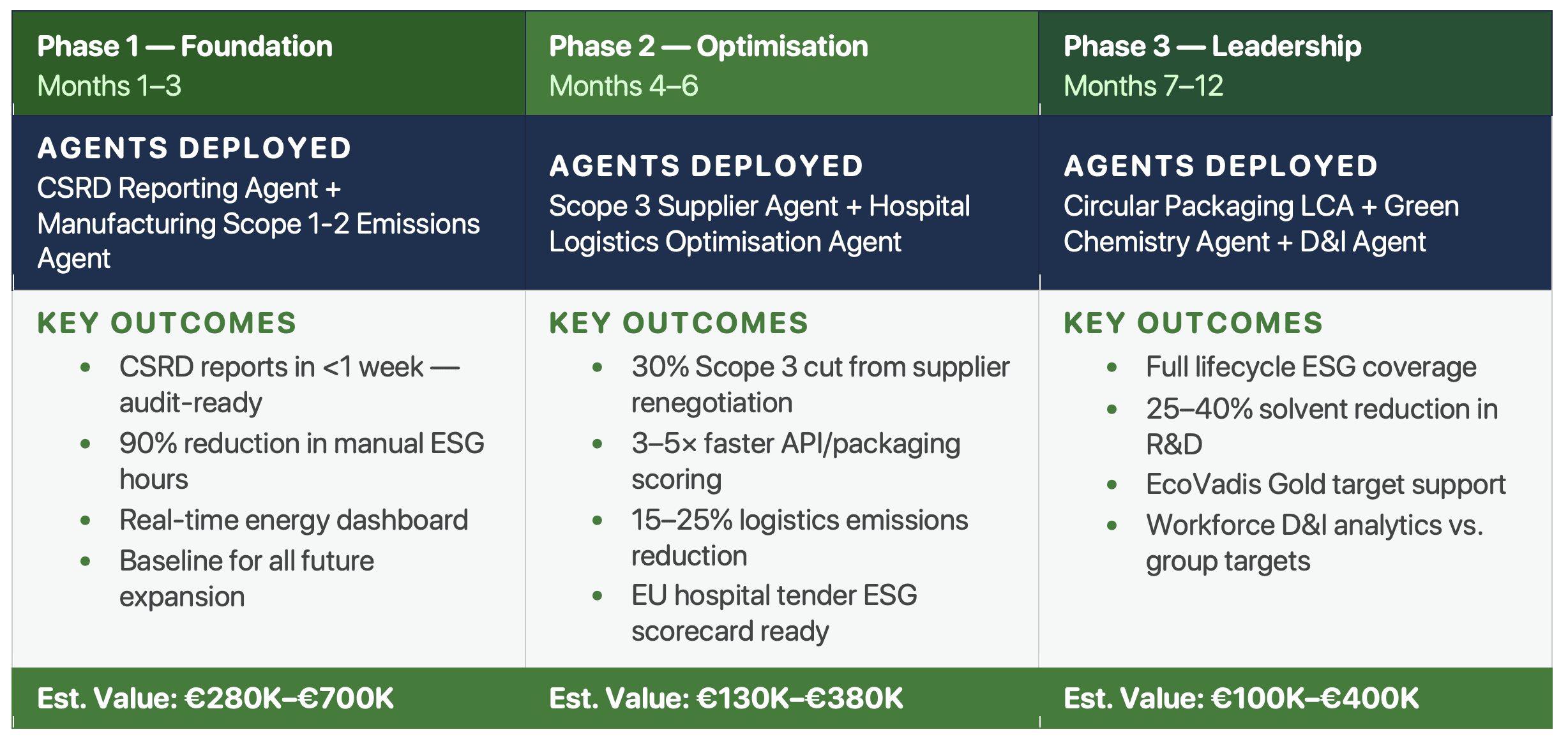
The Agentics Validation-First deployment framework runs in three phases over 7–12 months. Phase 1 (Months 1–3) deploys CSRD Reporting and Manufacturing Emissions agents. Phase 2 (Months 4–6) adds Scope 3 and Logistics agents. Phase 3 (Months 7–12) completes with Packaging, Green Chemistry, and D&I agents. Each phase proves ROI before the next is authorised.
The implementation approach is built around a core principle: Validation-First. Every phase of deployment must prove its return on investment before the next phase is authorised. There are no big retainers, no lock-in, and no consulting-style fee structures that reward hours over outcomes.
Section 08 - COMPETITIVE LANDSCAPE
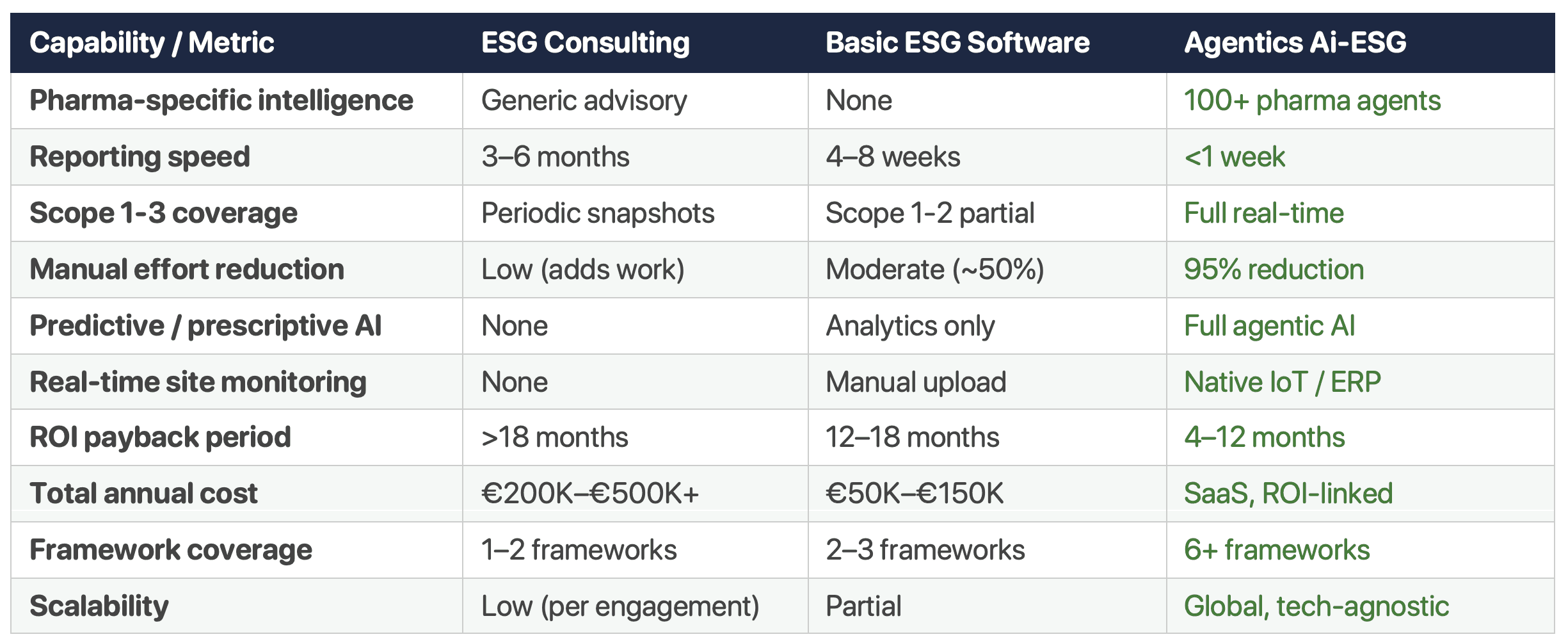
How Does Agentic Ai-ESG Compare to Traditional ESG Consulting and Software?
Agentics Ai-ESG platform outperforms traditional ESG consulting on speed (weeks vs months), coverage (6+ frameworks vs 1–2), effort reduction (95% vs low), and cost model (ROI-linked SaaS vs €200K–€500K+ retainers). They outperform basic ESG software on intelligence depth (100+ pharma agents vs generic templates), predictive capability (prescriptive recommendations vs analytics only), and integration (native IoT/ERP vs manual upload).
Section 09 - WHY THE AGENTS
Why Choose Agentics for Pharmaceutical Generics Ai-ESG?
Agentics, with its Ai-ESG Platform powered by Ecoratings.ai, is a leading Ai-ESG provider with 100+ pharmaceutical-specific agents, ISO 9001:2015 and ISO 14065:2020 certification, Verra Carbon Registry registration, a Validation-First ROI-linked engagement model, and proven deployment at organisations including Tech Mahindra, Accenture, M&S, and Soothe Healthcare.
01 Pharma-Native Intelligence — 100+ agents trained specifically on generics manufacturing, API supply chains, blister LCA, hospital logistics, and EU pharmaceutical regulatory frameworks. Not generic industry templates.
02 Results, Not Reports — We deploy self-learning AI agents that create measurable outcomes and put our fees at risk on the ROI we promise. Validation-First, phased engagement. No big retainers. No lock-in.
03 Audit-Ready from Day 1 — ISO 9001:2015 · ISO 14065:2020 · Verra Carbon Registry certified. All outputs formatted for direct submission to CSRD regulators, CDP, EcoVadis, and parent group reporting systems.
04 Global Platform, Local Execution — Presence in Amsterdam, Istanbul, Toronto, Bengaluru, Mumbai, Warsaw, Lisbon, and 5 further cities. EU pharma and Portuguese market expertise in a single delivery team.
05 Proven at Scale — Deployed at Tech Mahindra, Accenture, M&S, Soothe Healthcare, HMEL, LT Foods, and 20+ global organisations. Forbes India Select 200 · NVIDIA Inception · Microsoft for Startups.
06 Anti-Consultancy Model — We build and run AI systems, then hand them over. You own the platform. We charge for outcomes, not hours. One point of accountability across design, build, and run phases.
CONCLUSION
The Pharmacenutical Generics ESG Race Has Already Started
The pharmaceutical generics industry stands at an inflection point that will determine competitive positioning for the next decade. The forces driving ESG transformation i.e. CSRD, hospital procurement criteria, Scope 3 accountability, parent group reporting obligations, and the accelerating cost of manual compliance, are not future risks. They are present realities.
The companies that will lead this industry in 2030 are the ones investing now in AI-native ESG infrastructure. Not to tick compliance boxes. Not to satisfy investor questionnaires. But because they understand that sustainability data, properly captured and intelligently applied, is a lever for operational efficiency, market access, supply chain resilience, and talent attraction that compounds over time.

Proposed Next Steps
1. 30-min Discovery Call — Align on your ESG priorities, CSRD obligations, and platform fit. No commitment required.
2. Paid ESG Discovery Workshop — 1–2 day baseline audit + CSRD gap analysis + live ROI model with your actual operational data. Investment is credited against Phase 1 deployment fee.
3. Live Ai-ESG Platform Demo — See the platform running with your industry's data. No generic demos, no pre-recorded walkthroughs.
4. Phase 1 Pilot Deployment — Deploy the CSRD Reporting Agent and Manufacturing Scope 1-2 Emissions Agent. Proof of value within 90 days.
5. Phase 1 ROI Review & Phase 2 Go/No-Go — Joint steering committee at end of Month 3. Next phase authorised only on demonstrated results.
CONTACT US
Ready to evaluate Agentic AI for your ESG initiatives?
Email: Hello@TheAgentics.co
Website: https://TheAgentics.Co
WE BUILD. WE SCALE. WE OPTIMIZE.
Transform Your Pharmaceutical Business into an AI-Native Enterprise
Related Post

AI Transformation
Apr 14, 2026







